Angiogenesis and Inflammation
Prof. Michael Stürzl, PhD
with Jisheng Chen, MD, Richard Demmler, M.Sc.,Tobias Gass, Yanmin Lyu, MD, Mariam Mohamed Abdou, M.Sc., Katja Petter, Christina Schnürer, Chiara Van Passen, M.Sc., Luisa Weiss, MD, Taoyu Yang, MD.
The group develops novel strategies to map the temporal and spatial appearance of different endothelial cell activation phenotypes such as proliferation, invasion, migration and apoptosis both, under physiological and pathological conditions. Enlightenment of the molecular regulation of these phenotypes in inflammation-associated angiogenesis will enable us to modulate blood vessel activation with therapeutical intention.
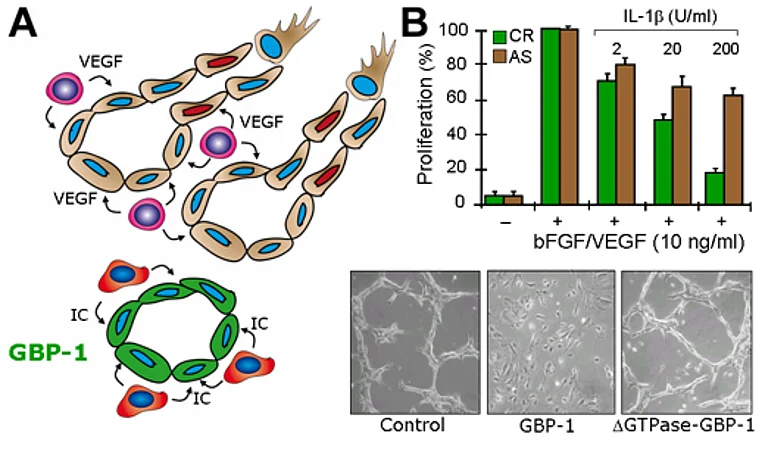
In previous work we have identified the guanylate binding protein-1 (GBP-1) as a novel marker of endothelial cells that are activated by inflammatory cytokines (IC) such as IFN-γ, IL-1β and TNF-α both, in vitro and in vivo. However, GBP-1 was not only a marker but also a key regulator of the antiangiogenic phenotype induced by IC. The protein was necessary and sufficient to mediate the inhibition of proliferation and invasion of EC triggered by IC. In addition, GBP-1 is the first GTPase that was found to be specifically secreted by endothelial cells and its concentration was increased in the cerebrospinal fluid of bacterial meningitis patients. This opens up perspectives to specifically detect the inflammatory activation of the blood vessel system in the serum of patients.
Recently, we were able to show that GBP-1 also participates in tumor biology. Angiogenesis and inflammation are prominent stroma reactions that are involved in the pathogenesis of colorectal carcinoma. Carcinomas expressing GBP-1 were characterized by an intrinsic immunoangiostatic tumor microenvironment. Most importantly, GBP-1 was found to be an independent prognostic factor indicating a risk of cancer-related death that is lowered by 50% in the colorectal carcinoma.